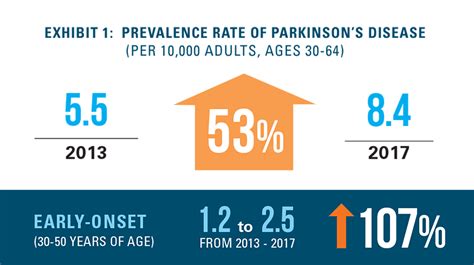
The etiology of Parkinson's disease is not yet clear. Parkinson's disease is widespread in all countries, without geographical or ethnic distinction. Parkinson's disease (PD) is second only to Alzheimer's in prevalence and represents one of the most complex neurological challenges of our time. In Italy, there are over 300,000 affected individuals. PD can no longer be classified as a motor disorder but as a complex pathological context characterized by a wide spectrum of non-motor symptoms (NMS), which can manifest in all stages of the disease, even up to 20 years before clinical diagnosis. These frequently include: constipation, anxiety, depression, apathy, pain, and sleep disturbances. Beyond the intensity with which each NMS can manifest, what impacts quality of life is the total number of NMS present in the patient. Therefore, recognizing them as early signs of the disease is crucial for undertaking a targeted and timely therapeutic path, intervening at a time when neurodegeneration has not yet reached a stage that causes motor symptoms.
Current pharmacological treatments are exclusively symptomatic and fail to meet the need for a multifactorial approach capable of addressing the broad spectrum of NMS. Within non-pharmacological therapy, nutraceuticals are positively emerging, offering new perspectives in supporting brain health. Among the most promising active substances, and those of growing scientific interest, are dry extract (e.s.) of green tea, titrated in epigallocatechin-3-gallate (EGCG), dry extract of saffron titrated in crocin and safranal, alpha-lipoic acid, omega-3 fatty acids, B vitamins, as well as probiotics and prebiotics. In particular, green tea dry extract has an antioxidant action, contributes to normal intestinal function, and acts as a tonic in cases of physical and mental fatigue. The intake of omega-3, on the other hand, is supported by clinical evidence demonstrating that, when combined with vitamin E, it can improve the quality of life of patients by reducing non-motor symptoms, as assessed by the UPDRS scale. Furthermore, docosahexaenoic acid (DHA), from the omega-3 series, is recognized for its ability to contribute to normal brain function. Saffron dry extract, titrated in crocin and safranal, is known for maintaining normal mood. Indeed, a dose of 30 mg/day is recommended by Canadian guidelines for the management of major depressive disorder. Finally, the intake of prebiotics and probiotics is useful for promoting the rebalancing of intestinal bacterial flora, which is significantly altered in subjects affected by PD. In light of the numerous research findings in this area and considering that clinical practice is increasingly oriented towards personalized care, nutraceuticals are configured as a valid complement to conventional therapy, offering natural, effective, and safe solutions.
KYMAX®, a food supplement from KOLINPHARMA S.p.A., an Italian nutraceutical company, fits into this scenario with an innovative and patented formulation, the result of years of research, aimed at the well-being of Parkinson's patients in their daily lives. The formulation of KYMAX®, containing DHA (DHA Origins®), alpha-lipoic acid (MATRIS®), green tea dry extract, saffron dry extract, vitamin E, B vitamins, L. rhamnosus LR-06, and inulin, was created with the aim of contributing to a more synergistic and personalized management of the patient, responding to the ever-increasing need for daily and complementary support to conventional therapy.
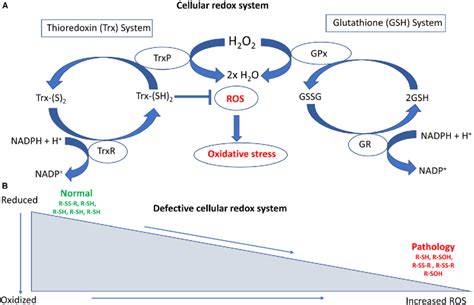
The redox system is a biochemical system present both outside and inside cells, consisting of a pool of chemical species that exchange electrons. This exchange of electrical charges constitutes the mechanism the system uses to regulate and defend the body, allowing it to respond optimally, adapt, and survive events. It is the link between the body's cells and the bacteria of the intestinal microbiota, capable of controlling the expression of numerous genes.
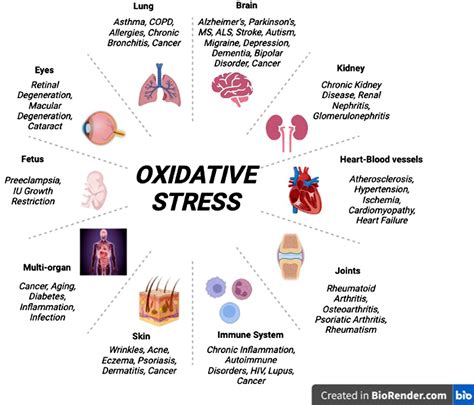
Exogenous factors of a physical nature such as radiation, of a chemical nature such as pollution, or biological such as infections, and endogenous factors such as a poor lifestyle, including emotional stress, smoking, vaping, alcoholism, and inadequate physical exercise, can cause oxidative stress. Situations in which a person under stress easily develops a cold sore or manifests inflammatory bowel diseases such as irritable bowel syndrome, or psoriasis, stress-related dermatitis, or even bone and joint pain in other locations, are well known. There are events and contexts that generate distress, an anglicism that identifies a continuous stimulation induced by an abnormal event that alters the normal biorhythm and generates a continuous inflammatory state with repercussions on health and well-being. Inflammation is not only a local phenomenon like an abscess but also systemic, such as a fever that affects the entire organism. While inflammation is a defense mechanism in its initial phase, continuous inflammation can support the onset of chronic or severe pathologies. To adapt to the action of oxidizing agents, cells develop various defense mechanisms: they regulate cellular metabolism, energy production, survival, death, and cell turnover. Common antioxidants constitute a heterogeneous family of substances that can be classified in various ways. Vitamin C contributes to the regeneration of the reduced form of vitamin E, and also contributes to the normal formation of collagen, the structural basis of blood vessels and skin, whose integrity is compromised by oxidation and inflammation, and is a contributing factor to events affecting the cardiovascular system. A greater effect in counteracting oxidative stress is given by the synergistic action of vitamin C, Selenium, Zinc, and vitamin E, without neglecting the fundamental effect of B vitamins necessary for normal energy metabolism and normal nervous system function. The effect of Magnesium is also relevant, as it has a muscle-relaxant action and is essential for the functioning of the heart and nervous system, and Omega-3s, which can fluidify blood and reduce inflammation levels. The study of oxidative stress allows not only to evaluate the state of health but also to correctly frame the patient for any preventive or curative intervention. The secret to living better and aging well, in addition to a bit of luck, therefore consists in keeping the Immune System trained and reducing inflammation levels by adopting an adequate lifestyle. This means, first and foremost, choosing what and how much to eat and how to supplement the diet. These choices have a decisive importance for health, for controlling inflammation, and influence the intestinal microbiota. Equally necessary is physical activity to control weight and reduce the risk of degenerative diseases such as Parkinson's and Alzheimer's. The level of alcohol consumption that minimizes the risk of health damage is, according to the latest guidelines, zero, as it is responsible for numerous diseases.
Congenital muscular dystrophy of Ullrich and Bethlem myopathy are rare hereditary muscle diseases caused by mutations in the COL6 genes (A1, A2, A3), for which a cure is not yet available. Studies in col6-deficient mice have revealed that muscle fiber degeneration involves autophagic defects and that forced activation of autophagy leads to improvement of the muscle pathology. Seven adult patients with myopathies caused by Col6 deficiency were subjected to a controlled low-protein diet for 12 months, and the presence of autophagosomes and levels of mRNA and protein for BECN1/Beclin 1 and MAP1LC3B/LC3B were evaluated in muscle biopsies and blood leukocytes. Muscle strength, motor and respiratory function, and metabolic parameters were assessed. After one year on a low-protein diet, autophagic markers increased in skeletal muscles and blood leukocytes of the patients. The safety of the treatment is demonstrated by the preservation of body composition fat percentage, muscle strength, and function.
Introduction: COL6/collagen VI is one of the main extracellular matrix proteins of the endomysium of skeletal muscles. Mutations in the COL6 genes (COL6A1, COL6A2, and COL6A3) cause COL6-related myopathies, a group of rare inherited muscle diseases that include Bethlem myopathy (BM) and Ullrich congenital muscular dystrophy (UCMD), as well as limb-girdle and myosclerotic variants of myopathy. BM is characterized by axial and proximal muscle weakness, along with contractures of the interphalangeal joints of the last 4 fingers. BM is usually mild and sometimes slowly progressive. Respiratory insufficiency is part of the clinical spectrum and can occur in ambulatory patients. UCMD is a severe congenital muscular dystrophy characterized by early onset, wasting and generalized muscle weakness, proximal joint contractures, and distal joint hyperflexibility. Independent walking is rarely achieved or maintained in children with UCMD, who also suffer from early and progressive respiratory insufficiency. We previously demonstrated that cyclosporine A can rescue muscle alterations associated with COL6 deficiency in diseased mice and in cells from BM or UCMD patients. Although clinical studies have indicated that cyclosporine A may be useful in slowing disease progression by correcting mitochondrial dysfunction, the well-known immunosuppressive activity of cyclosporine A is a major obstacle and hinders its long-term use, especially in pediatric patients. Recent studies in diseased mice and patient biopsies have provided new insights into the molecular mechanisms underlying COL6-related myopathies. These studies have shown that a failure of autophagic machinery plays an important pathogenetic role in muscle wasting and weakness. Autophagy is an evolutionarily conserved self-degradative process that is essential for nutrient recycling during fasting and for the removal of harmful or damaged cellular components. In skeletal muscles of diseased mice (col6a1), impaired autophagosome formation triggers the accumulation of dysfunctional mitochondria and organelles in myofibers, leading to apoptosis and myopathy. Specifically, restoring appropriate autophagic flux by genetic, pharmacological, or nutritional approaches rescues the myopathic phenotype of col6a1 mice. Here, we report the results of a pilot clinical trial with an LPD conducted for 12 months in 7 adult subjects with genetically characterized COL6 mutations (6 BM and 1 UCMD). The primary endpoints of this nutritional approach were successfully met, as the safety of the treatment was demonstrated and patients' muscles showed increased levels of the autophagic markers BECN1 and MAP1LC3B/LC3B after treatment. Furthermore, and more importantly, this biological response was accompanied by a lack of disease progression and improvement in some functional and metabolic parameters. In particular, the incidence of apoptosis in muscle biopsies was significantly reduced after the LPD diet. These results indicate that nutritional approaches can be effectively used to induce autophagy in patients with COL6-related myopathies and target autophagy induction as a valuable therapeutic target to improve muscle health in these patients. In a broader sense, our data provide a rationale for new treatment opportunities in an increasing number of muscle and non-muscle disorders associated with autophagic impairment.
Discussion: The results of this nutritional pilot trial, although conducted on a small number of patients with a rare disease, provide valuable insights for the medical community. Indeed, this is the first clinical study showing autophagic induction in a human hereditary disorder, with beneficial effects in counteracting disease progression. The improvement of multiple functional parameters, despite some degree of body weight loss, is a remarkable achievement, confirming the safety and potential benefits of autophagy induction in COL6-related myopathies. Given the early onset and progressive course of COL6-related myopathies, patients generally worsen over time, but not predictably. LPD treatment is expected to increase autophagy-mediated breakdown of proteins and organelles, and consequently, patients lose 7% of muscle mass. However, our data indicate that despite some degree of muscle mass loss and the fact that patients have aged, the absolute strength of leg muscles did not decrease after one year of LPD, and autophagy induction led to a decrease in myofiber apoptosis, improved motor function, and improved respiration. These seemingly paradoxical findings are readily explained by the critical control that autophagy exerts over the quality of proteins and organelles. By promoting autophagic flux, damaged organelles and proteins are eliminated, and muscles lose some of their mass but become better in function. Conversely, inhibition of autophagy in muscles is sufficient to induce wasting and weakness. It can be stated that the metabolic shift towards lipid oxidation that we observed in all patients after one year of LPD is based on improved mitochondrial function elicited by autophagy induction. This is consistent with the fact that fatty acid β-oxidation is one of the main energy metabolism pathways operating within mitochondria. The observation that serum lactate and acetate decreased after one year of LPD further points to more efficient mitochondrial oxidative activity. We emphasize that our data represent a "static" analysis of autophagy and cannot provide insights into autophagic flux due to the lack of appropriate methods for flux detection in humans. The limitations of this pilot clinical study are inevitably linked to the low prevalence of BM and UCMD and the small number of patients eligible for such a pilot trial. Indeed, it is extremely difficult to design randomized studies for patients with COL6-related myopathies or to include a standard dietary control group for LPD, due to obvious ethical constraints raised by multiple invasive muscle biopsies required to assess efficacy. Despite these inherent limitations, our findings are very promising and pave the way for nutritional interventions aimed at inducing autophagy in neuromuscular disorders. In reality, the objective of this study was to investigate the utility of targeting autophagy in COL6-related myopathies, thus providing the basis and justification for future larger clinical trials. It should be emphasized that this pilot clinical study is not intended to recommend LPD as a definitive long-term treatment for patients with BM or UCMD. Instead, our study aimed to evaluate whether autophagy can be induced in BM and UCMD patients and whether such induction is safe and has beneficial effects towards a future therapy. There is a growing need to identify biomarkers and tools to monitor autophagy in humans without using biopsies. Our work is the first study to demonstrate that blood leukocytes represent a promising non-invasive tool for assessing the autophagic status in neuromuscular diseases, both in animals and humans. This finding is particularly relevant for tissues not easily accessible by biopsy, such as the brain and heart, and in which autophagy is an important homeostatic player. Indeed, autophagy is a valuable and druggable target for combating neurodegenerative disorders, such as Huntington's, Alzheimer's, and Parkinson's diseases, as well as congenital or acquired cardiomyopathies. Overall, our data have met the objectives of this pilot LPD clinical trial, demonstrating not only that autophagy can be induced in myopathic patients but also that autophagy is a valuable target for novel therapeutic approaches. In the near future, the combination of natural compounds that modulate autophagy with specifically designed nutritional programs may allow their actions to be synergized and further improve the modest benefits obtained in this study from LPD.
R&D and Quality Specialist, Kolinpharma S.p.A.
The information provided on this site is for informational and educational purposes only. It is recommended to always consult your primary care physician or a specialist for any questions regarding a medical condition. BIO-CLOCK SRL disclaims any responsibility for any consequences arising from the use of the information contained on this forum and on the website www.hebio.it, www.forum.hebio.it, and on the page dedicated to the forum. Readers, especially those taking prescription or over-the-counter medications, should consult their doctor before undertaking any nutritional, supplementation, or lifestyle program.
The doctor Michela Barichella, head of the Simple Clinical Nutrition Unit of ASST Gaetano Pini-CTO, and other specialists (including Prof. The subjects undergoing supplementation showed significant improvements: in a 6-minute walk test, those who had taken the supplement walked 18 meters more, a distance 25% greater than untreated patients. During the study, vitamin 25 OH D was also dosed in both groups, both at admission and at discharge. This allowed for an additional finding, as highlighted by Dr. Barichella: "An evidence, if you will, secondary to the study is having scientific proof that during hospitalization, vitamin D levels drop drastically, because patients live indoors even during daylight hours."
First of all, levodopa has competitive absorption with meal proteins, so the protein portion should be shifted to the evening: at lunch, therefore, carbohydrates and vegetables, and in the evening a dinner based on meat, fish, eggs, cheese, or legumes. I have suffered from Parkinson's for years; currently, in addition to specific medications, on your advice, I am taking the 'probikene' probiotics you recommended some time ago. Can I currently add other supplements to slow down the disease? I also take vitamin D and vitamin C. Furthermore, I would like to ask you if those soluble collagen preparations are effective for improving skin tone, and if so, what do you recommend? If I consume the centrifuged juice of 3 carrots every day, does this raise my blood sugar? If so, what precaution should I take to avoid it? Is it true that eating fruit on an empty stomach raises blood glucose, and therefore it is better to consume it at the end of a meal?
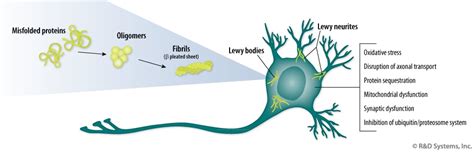
The phytochemicals previously mentioned boast among their biological effects: modulation of dopamine metabolism and release, growth factor induction, antioxidant and anti-inflammatory activity, regulation of mitochondrial homeostasis, modulation and neurogenesis of stem cells, restoration of correct protein folding mechanisms and autophagy. The figure illustrates the effects of plants rich in phytochemical compounds in counteracting the cascade of molecular events occurring in Parkinson's and parkinsonisms. These effects include: oxidative stress and accumulation of reactive oxygen species generated by altered dopamine metabolism; mitochondrial and endoplasmic reticulum stress; structural alterations of alpha-synuclein, a protein produced by neurons that regulates neurotransmitter traffic at the synaptic level; neuroinflammation; incorrect autophagy.
Recreating damaged nervous tissue from Parkinson's disease in the laboratory: a goal that could be possible starting from somatic cells. Scientists at the San Raffaele Scientific Research Institute have indeed identified three genes that, when activated, transform fibroblasts, the cells that provide fibers and collagen to tissues and ensure their structure, into induced dopaminergic neurons (iDA). The latter have functionalities entirely similar to those of the brain cells found in the area of the brain affected by the disease, called dopaminergic neurons. It was already known to researchers that neuron transplantation could be part of the treatment for neurodegenerative disease, but until now, the only source usable for clinical purposes for the production of brain tissue was embryonic stem cells. For this reason, the Milanese research group has developed a new technique based on the direct conversion of connective tissue cells from healthy donors or from Parkinson's patients themselves.
Effects of Collagen on Brain Structure and Cognitive Function
Dietary Vitamin E as a Protective Factor for Parkinson's Disease: Clinical and Experimental Evidence. Front Neurol. 2019 Feb 26;10:148. Titova N et al. Non-motor Parkinson disease: new concepts and personalized management. The Medical journal of Australia. 2018; 208(9): 404-409. Kalia LV et al. Parkinson’s disease. Lancet. 2015; 386: 896-912. Mostile G et al. Treatment of dysautonomia associated with Parkinson’s disease. Parkinsonism and Related Disorders. 2009; 5: s224-s232. Annex 1 to DM 10 August 2018. Li FJ et al. A meta analysis of tea drinking and risk of Parkinson's disease. Scientific World Journal. 2012;2012:923464. doi: 10.1100/2012/923464. Epub 2012 Feb 15. PMID: 22448141; PMCID: PMC3289976. Tamtaji O et al. The effects of omega-3 fatty acids and vitamin E co-supplementation on gene expression related to inflammation, insulin and lipid in patients with Parkinson's disease: A randomized, double-blind, placebo-controlled trial. Clin. Neurol. Neurosurg. 2019. Reg CE 432/2012. Sarris J et al. Clinician guidelines for the treatment of psychiatric disorders with nutraceuticals and phytoceuticals: The World Federation of Societies of Biological Psychiatry (WFSBP) and Canadian Network for Mood and Anxiety Treatments (CANMAT) Taskforce. World J Biol Psychiatry. 2022;23(6):424-455. An article review titled “Phytochemicals bridging autophagy induction and alpha-synuclein degradation in parkinsonism” (doi:10.3390/ijms20133274) has been published online in the journal International Journal of Molecular Science, which includes among its authors Maico Polzella, head of production and founder of Aliveda, with coordination by Professor Francesco Fornai, from the Department of Translational Research and New Technologies in Medicine and Surgery at the University of Pisa.